Forgot your password?
Background
A patent foramen ovale (PFO) is a persistent connection between the left and right atrium, affecting between 20-34% of the population (1), whereas in the majority of the population, this will close shortly after birth as left atrial pressure rises above right atrial pressure (2). Although a benign finding in most, PFOs can have important clinical relevance, allowing the passage of material from the venous to the arterial circulation, referred to as paradoxical embolism (3), and it is in stroke where this becomes most relevant. Importantly, paradoxical embolism is believed to account for up to 5% of all strokes, and up to 10% of stroke in younger patients (4), but crucially, a large number of ischaemic stroke remain referred to as ‘cryptogenic’, with no clear cause identified, with suspicion that the burden of thromboembolic stroke is being underestimated (5). Therefore, the idea of closing the PFO as a mechanism of preventing recurrent embolic stroke, or to give reassurance to young stroke patients that one key potential cause of stroke has been eliminated is highly appealing.
Evidence for Closure
RCT data was initially inconclusive for whether percutaneous PFO closure reduces recurrent embolic events. The PC Trial, using the AMPLATZER device demonstrated safety of the device, but did not show any significant reduction in recurrent embolic events or death in patients with cryptogenic stroke and a confirmed PFO, for PFO closure compared with medical therapy. This comprised 204 patients in the closure group and 210 with medical therapy over a mean follow-up of 4.1 years (6). The CLOSURE I trial, using the STARFlex system, enrolled 909 patients and had a composite primary endpoint of stroke or TIA in two-years of follow-up, all-cause mortality in the first thirty-days, and death from neurological cause between thirty-days and two-years. This trial again failed to demonstrate benefit of device PFO closure compared with medical therapy (7). Results became more encouraging for device PFO closure as time progressed, the RESPECT trial, using the AMPLATZER device, included 980 cryptogenic stroke patients, with a median follow-up of 5.9 years, and did show a reduction in rates of recurrent ischaemic stroke when compared to medical therapy alone (8). The CLOSE trial randomised 663 patients with recent stroke attributed to PFO, with an additional associated atrial septal aneurysm or large interatrial shunt, to PFO closure with antiplatelet therapy or antiplatelet therapy alone, and showed a reduction in rate of stroke recurrence in the PFO closure arm (9). Similarly, the REDUCE trial included 664 patients with cryptogenic stroke, 81% of whom has moderate or large interatrial shunts, and demonstrated lower risk of recurrent ischaemic stroke in the PFO closure arm, although did note the increased risk of device complications and atrial fibrillation in patients undergoing PFO closure(10).
Following this, there have been a range of meta-analyses that have shown consistent benefit for device closure. Kent et al conducted the first pooled analysis of individual participant data from the three previously mentioned trials, showing a reduction in recurrent stroke, TIA or all-cause mortality after covariate adjustment (11). Khan et al with a similar meta-analysis demonstrated a similar benefit for device closure (12). Finally, Rengifo-Moren et al suggested that device-closure of PFO could reduce risk of recurrent vascular events (including stroke, TIA and peripheral embolism), and interestingly raised that the benefit could be greater in those with a substantial shunt (13).
Who should have their PFO closed?
The BCIS position statement from 2025 identifies the following groups of patients in whom the diagnosis of a PFO should be pursued, importantly noting that the diagnosis of PFO is of importance only where closure would be indicated (14).
The main source of referral for PFO closure is from stroke teams, with a structured approach taken to investigations after stroke in younger patients without obvious risk factors. From a cardiovascular perspective, patients are often investigated with a 12-lead ECG and/or a period of Holter monitoring, then progress to transthoracic echocardiogram and bubble contrast echocardiogram if there is no evidence of atrial fibrillation (15). Cardiologists are often then meeting patients for the first time in the outpatient department in the weeks following ischaemic stroke, typically after a positive bubble echocardiogram and the absence of an alternate cause of stroke in the rest of the ‘young stroke’ work-up.
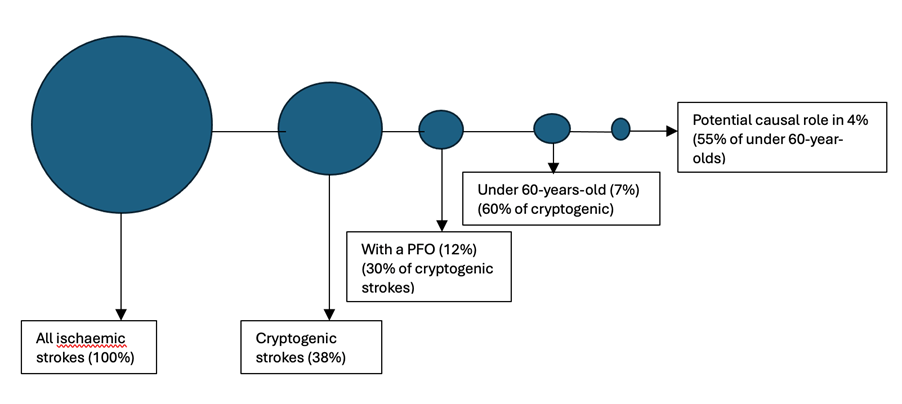
Figure 1: Proportion of ischemic stroke with patent foramen ovale (PFO) as a potential cause. Proportions are derived from a cohort of 15 239 patients with ischemic stroke from the London Ontario Stroke Registry with complete echocardiograms(3)
Figure recreated from: Sposato LA, Albin CSW, Elkind MSV, Kamel H, Saver JL. Patent Foramen Ovale Management for Secondary Stroke Prevention: State-of-the-Art Appraisal of Current Evidence. Stroke. 2024 Jan;55(1):236-247. doi: 10.1161/STROKEAHA.123.040546. Epub 2023 Nov 21. PMID: 38134261.
Figure 1 illustrates why PFO closure is becoming an important part of post-stroke care. With a significant proportion of strokes labelled as cryptogenic (approximately 25%), 16% can be further labelled as embolic stroke of undetermined source (16), and approximately one quarter of these patients will have a PFO (3). There is therefore growing consensus to label these strokes as PFO-associated stroke rather than cryptogenic(17).
Are all PFOs equal?
It is also important to consider that not all PFOs are the same, and that there are features that modify the risk of stroke. Size of PFO matters, as quantified by number of bubbles that cross into the left atrium during agitated saline challenge within three cardiac cycles after opacification of the right atrium (3). The numbers of bubbles seen in the left atrium required to indicate ‘Large’ PFO ranges between 20-30 typically (18). Atrial septal aneurysm (ASA) refers to a deformity of the interatrial septum, protruding into either the right or left atrium, and typically arises at the level of the fossa ovalis (19). Pooled meta-analysis data showed that patients with a large shunt and atrial septal aneurysm gained substantially greater benefit with PFO closure than those without, identifying this as a particularly high-risk patient group (20).
How?
PFO closure is undertaken as a day-case procedure and can be guided by transoesophageal echocardiography (TOE) under a general anaesthetic, or with conscious sedation and intracardiac echocardiography (ICE). The BCIS position statement recommends ultrasound guidance for the femoral puncture and following puncture 70-100IU/kg of heparin to be given. The PFO is typically crossed with a J-tipped guidewire and a 6FR multipurpose catheter, which is then ideally placed in the left upper pulmonary vein, guided by the imaging modality of choice. Preferred modes of device size selection can vary, including usage of sizing balloons and echocardiography-guidance (either ICE or TOE). Once sized, the device is deployed using a delivery sheath, which is advanced via the IVC into the right atrium, through the PFO. Firstly, the left atrial disk of the device is expanded, this is retracted back towards the septum, and the delivery sheath is retracted, deploying the right atrial disk. The device will now be deployed, location can be checked with the imaging modality of choice, and then the delivery sheath removed (21).
Registrar and Fellow involvement
Although exposure to performing these procedures can be limited in training, many of us will rotate to centers performing these procedures and look after patients’ post-procedure. Although complications are rare (<1/100 cases), it is still important to be aware of the major contributors:
In the medium term, there appears to be an increased risk of atrial fibrillation post device insertion, arising in between 2.5-5% of patients (22). Although the mechanism of this is not entirely clear, this does appear to be transient and benign, often spontaneously resolving within forty-five days, requiring an individualized approach to management, and is not typically anticoagulated (23). Other considerations revolve around decisions regarding antiplatelet therapy, which will typically be for a minimum of 6 months (single or dual depending on operator), and then a decision regarding long-term antiplatelet therapy is typically taken according to patient risk-factors. BCIS recommend a single echocardiogram, which can be as an outpatient or pre-discharge, to assess device-position. Many centres perform a bubble echocardiogram at 3-6 months post-procedure to look for a residual shunt. The benefit of doing this is uncertain.
Conclusions
PFO device closure is a safe, well-tolerated procedure that is being carried out in increasing numbers. The evidence-base for PFO closure preventing recurrent embolic events following cryptogenic stroke is now strong. We have an important role, including within our general medical practice, to identify eligible patients and ensure prompt referral, and to be able to discuss the procedure with patients in our general cardiology clinic. Finally, although rare, it is important to be aware of the complications of PFO closure for those doing general cardiology on-call work, as these can be life-threatening events that require prompt recognition and management.
References