R&D Literature Review: DECEMBER 2025
BCIS literature review
December 2025
Authors: Dr. Natalia Briceno, Dr. Andrew Chapman, Dr. Richard Jabbour
VALVULAR HEART DISEASE
PARTNER 3: Transcatheter or Surgical Aortic-Valve Replacement in Low-Risk Patients at 7 Years
Rationale:
- There is a paucity of long-term comparative data on clinical outcomes and valve durability following transcatheter aortic-valve implantation (TAVI) versus surgical aortic-valve replacement (SAVR) among patients with severe symptomatic aortic stenosis deemed to be at low surgical risk.
- Prior analyses at 1 and 5 years have largely demonstrated similar outcomes
- This study aimed to assess 7-year outcomes, including composite clinical endpoints, valve performance, and durability which is especially important in younger lower risk patients
Publication Date:
- Leon MB et al. Transcatheter or Surgical Aortic-Valve Replacement in Low-Risk Patients at 7 Years. N Engl J Med. 2025 Oct 27. doi: 10.1056/NEJMoa2509766. Epub ahead of print. PMID: 41144631. October 27, 2025 (New England Journal of Medicine) DOI: 10.1056/NEJMoa2509766
- gov: NCT02675114
- Funding: Edwards Lifesciences
Study Design:
- Type: Prospective, randomized, multicenter, open-label clinical trial
- Trial: PARTNER 3
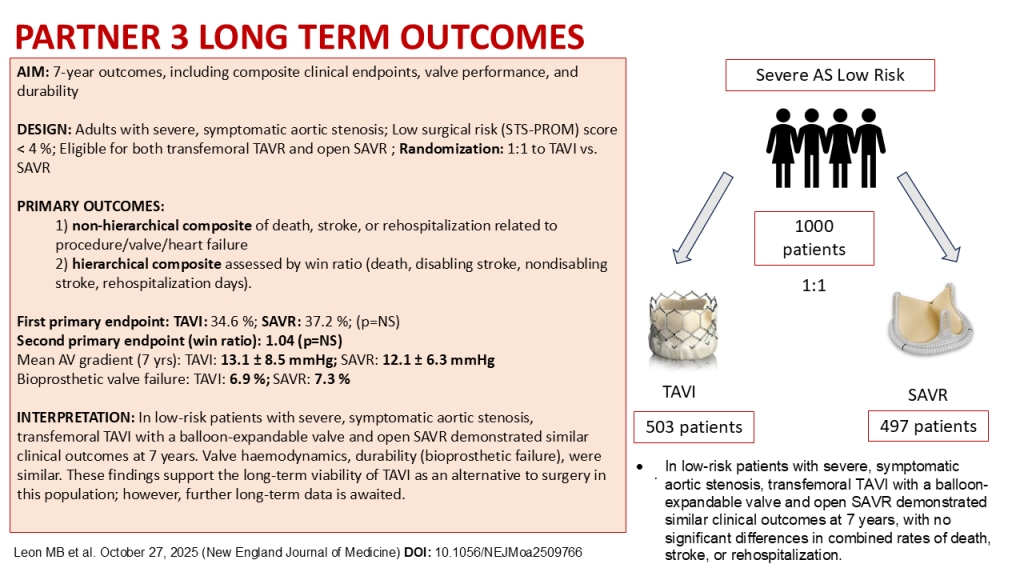
- Randomization: 1:1 to TAVI vs. SAVR
- Follow-up: 7 years
- Endpoints:
- Two primary endpoints
- 1) non-hierarchical composite of death, stroke, or rehospitalization related to procedure/valve/heart failure
- 2) hierarchical composite assessed by win ratio (death, disabling stroke, nondisabling stroke, rehospitalization days).
- Clinical, echocardiographic, durability, and health-status measures were also prospectively analysed
Inclusion Criteria:
- Severe, calcific aortic stenosis
- New York Heart Association Functional Class ≥ 2 OR exercise tolerance test that demonstrates a limited exercise capacity, abnormal BP response, or arrhythmia OR asymptomatic with Left Ventricular Ejection Fraction (LVEF) <50%
- Heart team agrees the patient has a risk of operative mortality and has an Society of Thoracic Surgeons (STS) score < 4
- The study patient has been informed of the nature of the study, agrees to its provisions and has provided written informed consent.
Selected Exclusion Criteria:
- Native aortic annulus size unsuitable for sizes 20, 23, 26, or 29 mm transcatheter heart valve
- Iliofemoral vessel characteristics that would preclude safe passage of the introducer sheath
- Evidence of an acute myocardial infarction ≤ 30 days before randomization
- Aortic valve is unicuspid, bicuspid, or non-calcified
- Severe aortic regurgitation (>3+)
- Severe mitral regurgitation (>3+) ≥ moderate stenosis
- Pre-existing mechanical or bioprosthetic valve in any position
- Complex coronary artery disease:
- Unprotected left main coronary artery
- Syntax score > 32
- Heart Team assessment that optimal revascularization cannot be performed
Characteristics:
- Total enrolled: 1000 patients
- TAVR group: ~503
- SAVR group: ~497
- Average age: ~73 years
- Mean STS score: ~1.9 %
Primary Endpoint:
- Non-hierarchical composite: death from any cause, stroke, or rehospitalization related to procedure/valve/heart failure
- Hierarchical composite: death, disabling stroke, nondisabling stroke, and number of rehospitalization days — analyzed using a win ratio
Results:
Primary Endpoints at 7 Years
- First primary endpoint (Kaplan-Meier):
- TAVI: 34.6 %
- SAVR: 37.2 %
- Difference: −2.6 pp (95 % CI, −9.0 to 3.7) — no significant difference
- Second primary endpoint (win ratio): 1.04 (95 % CI, 0.84–1.30) — no significant difference
Individual Components
- All-cause mortality:
- TAVR 19.5 % vs. SAVR 16.8 %
- Stroke:
- TAVR 8.5 % vs. SAVR 8.1 %
- Rehospitalization:
- TAVR 20.6 % vs. SAVR 23.5 %
Additional Outcomes:
- Valve Function & Durability:
- Mean aortic-valve gradient (7 yrs):
- TAVI: 1 ± 8.5 mmHg
- SAVR: 1 ± 6.3 mmHg
- Bioprosthetic valve failure:
- TAVI: 9 %
- SAVR: 3 %
- Patient-Reported Outcomes
- Quality-of-life measures showed no significant difference between groups at 7 years follow up.
- Mean aortic-valve gradient (7 yrs):
Limitations:
- Follow-up completeness: ~89 % at 7 years — late data lost to follow-up possible.
- Population: Low-risk cohort limits generalizability to moderate/high-risk or younger patients.
- Bicuspid, non-calcified valves excluded
- Some secondary outcomes with low events (e.g., pacemaker rates, paravalvular leak) were underpowered so limited conclusions can be made.
- Only performed in Sapien S3 Valves
Conclusions:
- In low-risk patients with severe, symptomatic aortic stenosis, transfemoral TAVI with a balloon-expandable valve and open SAVR demonstrated similar clinical outcomes at 7 years, with no significant differences in combined rates of death, stroke, or rehospitalization.
- Valve haemodynamics, durability (bioprosthetic failure), and patient-reported outcomes were comparable between approaches.
- These findings support the long-term viability of TAVI with the Sapien platform as an alternative to surgery in this population; however, further long-term data is awaited.
Percutaneous transcatheter valve replacement in individuals with mitral regurgitation unsuitable for surgery or transcatheter edge-to-edge repair: a prospective, multi-country, single-arm trial
Rationale
- To evaluate 1-year outcomes of a novel percutaneous transseptal transcatheter mitral valve replacement (TMVR) system in patients unsuitable for surgery or TEER.
Publication Date
Guerrero et al. Lancet. 2025; 406:2541-2550 November 29th, 2025.
Study Design
- Prospective, multicentre, single-arm trial
Population
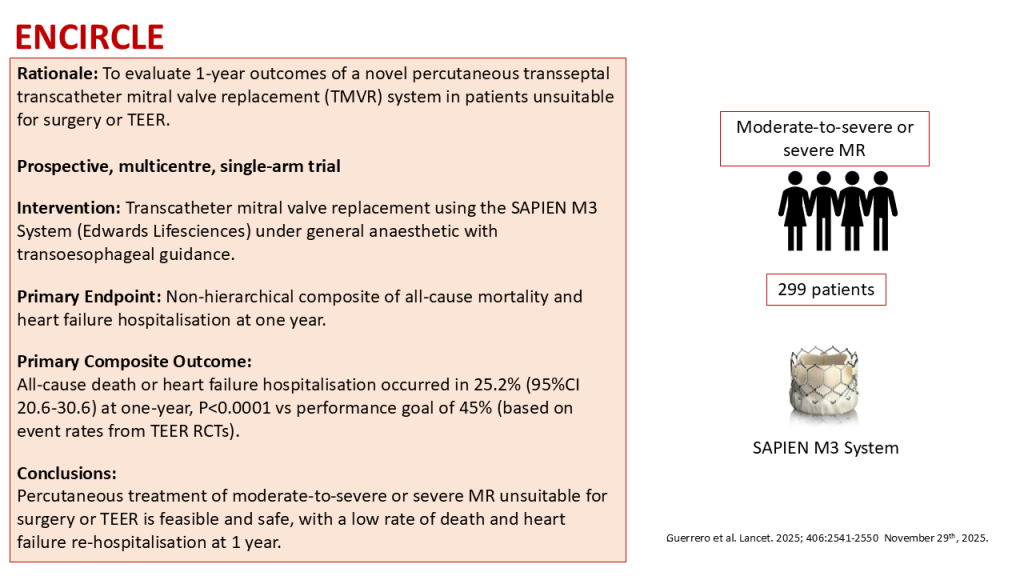
- Patients with symptomatic moderate-to-severe or severe mitral regurgitation (MR) who were not suitable for surgery or transcatheter edge to edge repair from 56 centres in six countries (USA, Canada, UK, Netherlands, Israel, Australia)
Intervention
- Transcatheter mitral valve replacement using the SAPIEN M3 System (Edwards Lifesciences) under general anaesthetic with transoesophageal guidance.
- This device uses a fully retrievable nitinol dock which encircles and captures the subvalvular apparatus, creating a landing zone for the balloon expandable transcatheter heart valve.
Control
- No control arm. Evaluated against a performance goal of 45%. This was defined based on event rates from trials of TEER against medical therapy and in conjunction with the US Food and Drug Administration.
Primary Endpoint
- Non-hierarchical composite of all-cause mortality and heart failure hospitalisation at one year.
Selected secondary endpoints
- Cardiovascular mortality. Heart failure hospitalisation. Improvement in MR. Improvement in NYHA class. Stroke. Clinically significant LVOT obstruction induced by SAPIEN M3. Mitral valve re-intervention. Paravalvular leak closure. Surgical MVR. Bleeding. Major structural complications. Major access site complications. Major iatrogenic septal defects.
Funding
- Edwards Lifesciences
Trial Info
ClinicalTrials.gov, NCT04153292
Inclusion Criteria:
Patients aged 18 years or over
NYHA functional class ≥2 and core lab moderate-to-severe or severe MR
Unsuitable for surgical or commercially available transcatheter treatment based on anatomical criteria or co-morbidity
Favourable anatomical features for treatment
– healthy leaflet tissue without perforation
– LV end diastolic diameter of 35-70mm
– commissure-commissure distance of <45mm
– absence of features associated with high risk of PVL, LVOT obstruction or difficulty implanting the dock or transcatheter heart valve
Anatomical suitability was determined using transthoracic and transoesophageal echocardiography and cardiac CT. Eligibility was confirmed by an independent physician screening committee.
Selected Exclusion Criteria:
– LV end diastolic diameter >75mm
– mitral annular or leaflet calcification that would interfere with device
– increased risk of PVL
– severe LV or RV dysfunction
– high risk of LVOT obstruction
Results:
A total of 1,171 patients were screened.
846 patients failed screening (410 anatomical exclusion, 280 inclusion criteria failed, 156 clinical exclusion)
299 patients treated (with 287 valves implanted; 96%). In 5 patients there were encircling difficulties, 5 had a low dock position, 1 had a pre-existing mobile echodensity and in 1 patient medial commissural access was not possible.
Primary Composite Outcome:
All-cause death or heart failure hospitalisation occurred in 25.2% (95%CI 20.6-30.6) at one-year, P<0.0001 vs performance goal
Additional Outcomes:
No difference between primary versus secondary MR
No patients required a second valve or conversion to surgery.
No intraprocedural deaths. Two in-hospital deaths and four in-hospital stroke events.
30-day outcomes:
Stroke (2.7%)
MV re-intervention (2.3%)
PVL closure (1.7%)
Clinically significant valve thrombosis (2.3%)
Haemolysis (4.3%)
New onset AF (7.9%)
Permanent pacemaker (2.6%)
1-year outcomes:
Stroke (2.7%)
MV re-intervention (2.3%)
PVL closure (1.7%)
Clinically significant valve thrombosis (2.3%)
Haemolysis (4.3%)
New onset AF (7.9%)
Permanent pacemaker (2.6%)
Sustained reduction in MR (grade 1 or lower) (96%)
Limitations:
1. Single arm study with no contemporary medical therapy control arm
2. Performance goal (45% primary endpoint) defined based on secondary MR patients in two prior RCTs of TEER therapy
3. Case selection bias with numerous exclusion criteria- findings reflect performance in optimal patient group selected with greatest likelihood of benefit
4. Only have outcomes to 1-year, however, inoperable patient cohort not suitable for TEER therapy therefore results are encouraging.
5. Risks of survival bias in symptom-based endpoints.
6. Performance in centres of expertise and real-world performance may differ
Conclusions
- Percutaneous treatment of moderate-to-severe or severe MR unsuitable for surgery or TEER is feasible and safe, with a low rate of death and heart failure re-hospitalisation at 1 year.
CORONARY ARTERY DISEASE
β Blockers after Myocardial Infarction with Mildly Reduced Ejection Fraction: An Individual Patient Data Meta-Analysis of Randomised Controlled Trials
Rationale:
- Evidence for the benefit of β-blocker therapy after acute myocardial infarction (MI) is well established in patients with reduced left ventricular ejection fraction (LVEF < 40 %).
- However, the benefit in patients with mildly reduced LVEF (40–49 %) but without heart failure remained uncertain because individual trials were underpowered for this subgroup.
- This study aimed to clarify whether β-blocker therapy reduces major adverse outcomes in this specific population.
Publication Date:
- Rosselló et al. β blockers after myocardial infarction with mildly reduced ejection fraction: an individual patient data meta-analysis of randomised controlled trials. The Lancet; 406(10508), September 13, 2025, Pages 1128–1137
DOI: 10.1016/S0140-6736(25)01592-2
Study Design:
- Type: Individual patient-data meta-analysis
- Data Source: Four recent randomized controlled trials
- REBOOT
- BETAMI
- DANBLOCK
- CAPITAL-RCT
- Population: Patients with acute MI (randomisation within 14days), LVEF 40–49 %, and no prior history or clinical signs of heart failure
- Intervention: β-blocker therapy versus no β-blocker therapy
- Model: One-stage fixed-effects Cox proportional hazards regression
- Primary Endpoint: Composite of all-cause death, new MI, or heart failure
- All endpoints independently adjudicated
Trial Info:
- Includes patients from multiple international RCTs that randomized MI patients within 14 days to receive β-blockers versus control
- Meta-analysis registered on PROSPERO (CRD420251023480)
Inclusion Criteria:
- Recent acute myocardial infarction
- Left ventricular ejection fraction (LVEF) 40–49 %
- Randomisation to β-blocker or control within 14 days post-MI
- No history or clinical signs of heart failure
Selected Exclusion Criteria:
- Prior clinical heart failure or signs thereof
- Contraindications to β-blocker therapy
Characteristics:
- Total patients analysed: 1,885
- β-blocker group: 991 patients (53 %)
- Control group (no β-blocker): 894 patients (47 %)
- Trials included (patients with mildly reduced EF):
- REBOOT: 979
- BETAMI: 422
- DANBLOCK: 430
- CAPITAL-RCT: 54
Primary Endpoint:
- Composite of:
- All-cause death
- New myocardial infarction
- Heart failure
Measured as events per 1,000 patient-years and hazard ratios via Cox regression.
Results:
Primary Composite Outcome
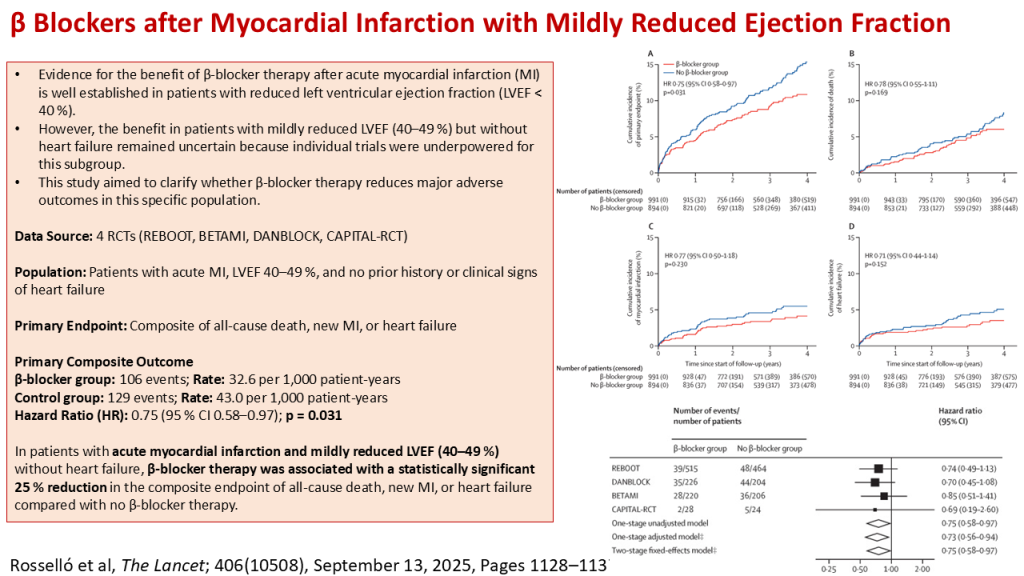
- β-blocker group: 106 events
- Rate: 32.6 per 1,000 patient-years
- Control group: 129 events
- Rate: 43.0 per 1,000 patient-years
- Hazard Ratio (HR): 0.75 (95 % CI 0.58–0.97); p = 0.031
- No heterogeneity: trial-by-treatment pinteraction = 0.95; country pinteraction = 0.98
This indicates a 25 % relative reduction in the risk of the primary composite outcome with β-blocker therapy.
Additional Outcomes:
- Consistent Directionality:
- Individual components (all-cause death, recurrent MI, heart failure) trended lower with β-blockers
- Heterogeneity:
- No significant differences between trials or across countries — indicating consistent effect across settings.
Limitations:
- Population: Focused only on patients without overt heart failure and mildly reduced EF; does not address preserved EF (≥50 %) or those with prior heart failure history.
- Median follow-up across included trials was >1 year
- Trial Differences: As an IPD meta-analysis pooling several trials, variations in β-blocker type, dosing, and other management may exist.
- In 3 trials (REBOOT, BETAMI, and DANBLOCK), they were pragmatically designed in nature, and therefore the categorisation of LVEF was based on data reported by the enrolling centres, which might have resulted EF misclassification
- All trials were open label which could introduce bias
Conclusions:
- In patients with acute myocardial infarction and mildly reduced LVEF (40–49 %) without heart failure, β-blocker therapy was associated with a statistically significant 25 % reduction in the composite endpoint of all-cause death, new MI, or heart failure compared with no β-blocker therapy.
- This outcome extends the recognised benefit of β-blockers from patients with reduced LVEF (<40 %) into this mildly reduced EF subgroup and supports continued use of β-blocker therapy in this population.
- This paper nicely complements another recent meta-analysis of 5 RCTs (1) with a total of 17,801 patients (REBOOT (7459 patients), REDUCE-AMI (4967 patients), BETAMI (2441 patients), DANBLOCK (2277 patients), and CAPITAL-RCT (657 patients) trials) with normal EF post MI which did not reduce the incidence of death from any cause, myocardial infarction, or heart failure in patients with an LVEF of at least 50% after myocardial infarction without other indications for beta-blockers.
- During a median follow-up of 3.6 years, a primary-end-point event occurred in 717 patients (8.1%) in the beta-blocker group and 748 patients (8.3%) in the no-beta-blocker group (hazard ratio, 0.97; P = 0.54).
- All-cause mortality: 335 patients (beta-blocker group) and 326 patients (no-beta-blocker) (hazard ratio, 1.04; 95% CI, 0.89 to 1.21)
- Myocardial infarction: 360 (beta-blocker group) and 407 patients (hazard ratio, 0.89; 95% CI, 0.77 to 1.03)
- Heart failure: 75 (beta-blocker) and 87 patients (hazard ratio, 0.87; 95% CI, 0.64 to 1.19).
- Kristensen AMD, et al. Beta-Blockers after Myocardial Infarction with Normal Ejection Fraction. N Engl J Med. 2025 Nov 9. doi: 10.1056/NEJMoa2512686. Epub ahead of print. PMID: 41211954.
Stratified treatment of myocardial infarction with non-obstructive coronary arteries: the PROMISE trial

Rationale: To determine if stratified treatment for myocardial infarction with non-obstructive coronary arteries improves clinical outcomes as compared to standard care
Publication Date: Montone RA et al. Eur Heart J. 2025. https://doi.org/10.1093/eurheartj/ehaf917
Study Design:
- Type: Multi-centre randomised controlled trial (1:1)
- Population: Patients with MINOCA
- Intervention: Comprehensive diagnostic work up to identify the underlying aetiology and target treatment
- Comparator: Standard care defined by the treating clinician
- Primary Endpoint: Between group difference in the change in angina status at 12-months assessed by Seattle Angina Questionnaire
- Secondary Endpoint: MACE (all-cause mortality, MI, stroke, HF hospitalisation and repeat angiography).
- All endpoints independently adjudicated
Trial Info:
Inclusion Criteria:
1. Myocardial infarction according to the Fourth Universal Definition criteria
2. No obstructive coronary artery disease (no stenosis >50%)
Selected Exclusion Criteria:
1. Contraindication to CMRI
2. End-stage renal or hepatic disease
3. Co-morbidity with life-expectancy of <1 year
4. Presence of non-cardiac or cardiac non-coronary causes of MI
5. Takotsubo cardiomyopathy
6. Clear aetiology of MINOCA evident at baseline angiography
Procedures:
Randomisation performed immediately after invasive angiography
CMRI performed in 80% of stratified treatment group and 75% of standard care
OCT performed in 43/45 (95.5%) of stratified treatment group
Acetylcholine provocation in 37/45 (82%) of stratified treatment group
Echocardiography in 3/45 (7%) of stratified treatment group
OCT and ACh not permitted in the standard care arm.
Results:
Primary Outcome:
At 12-months, SAQSS was significantly higher in the stratified treatment group vs standard care group (+9.38 (6.81 to 11.95, P<0.001).
Additional Outcomes:
Stratified investigation led to identification of cause of MINOCA in 80%.
Strengths:
– Demonstrating the feasibility of recruitment and randomisation of MINOCA patients in a multi-centre randomised trial is helpful
– Very detailed phenotyping with clear evidence of use of adjunctive diagnostic testing
Limitations:
– Small trial of 100 patients
– Unblinded treatment intervention with high risk of bias
– Early termination due to recommendation of the Data Safety and Monitoring Board – Primary outcome in favour of treatment but this was symptom based with no placebo control
– A very limited change in overall medical therapy received between treatment arms (the only significant difference being in the administration of non-dihydropyridine calcium channel blockers (33% [15/45] vs 2% [1/47]).
Conclusions:
Stratified treatment based on comprehensive diagnostic assessment and aetiology-guided therapy led to an improvement in angina-related health status in this small unblinded trial, but only a small difference in overall treatment was observed.
iModern: Immediate or Deferred Nonculprit-Lesion PCI in Myocardial Infarction
Rationale
- To investigate whether immediate iFR guided PCI of non-culprit lesions in patients presenting with STEMI and multi vessel coronary disease is superior to a deferred cardiac stress perfusion MRI guided approach.
Publications details:
- Nijveldt et al; iMODERN Investigators. Immediate or Deferred Nonculprit-Lesion PCI in Myocardial Infarction. N Engl J Med. 2025 Oct 28. doi: 10.1056/NEJMoa2512918. Epub ahead of print. PMID: 41159879.
Study design
- International, investigator initiated.
- Prospective open label randomised controlled superiority trial.
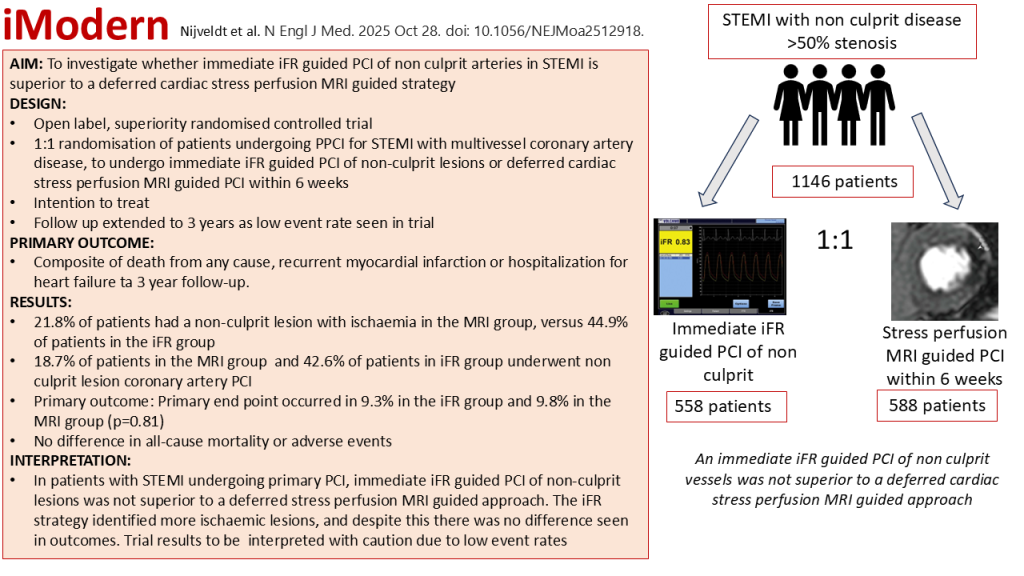
- 1:1 randomization of patients presenting with STEMI to undergo immediate iFR guided PCI of non-culprit lesions (defined as lesions with >50% stenosis and iFR less than or equal to 0.89) or deferred cardiac stress MRI guided PCI within 6 weeks after randomization.
Trial Information
- 1146 patients (558 in immediate iFR group and 588 in deferred MRI group)
- 41 sites
- Industry supported.
- COVID pandemic resulted in a pause to recruitment.
- Recruitment commenced in December 2017 and was complete in February 2022
- In the iFR arm, all non-culprit vessels were interrogated with iFR that had a diameter stenosis >50%. In those with a diameter stenosis of greater than 90%, PCI without iFR was allowed. PCI performed in all non-culprit lesions with an iFR of 0.89 or below. PCI of non-culprit could be performed within 24 hours.
- In the MRI arm, MRI performed within 6 weeks and angiogram and MRI images were analysed in a core laboratory for the adjudication of perfusion defects. A deferred iFR guided PCI procedure was allowed as a bail out.
- During trial recruitment there was lower than anticipated event rates, therefore follow up was increased to 3 years.
- Follow up 3, 6 and 12 months, and then at 3 years (middle 3 time points in person)
Inclusion
- 18 years or older
- Presenting as STEMI having undergone successful primary PCI of culprit artery
- PCI within 12 hours of symptom onset
- One or more non culprit lesions in a non-infarct related artery with a diameter stenosis greater than 50%
Exclusion
- Prior STEMI
- Chronic total occlusion
- Complex non culprit lesions
- Greater than 50% left main stenosis
- Cardiogenic shock
- Haemodynamic instability
- Acute pulmonary oedema
Outcomes
- Primary end point: composite of death from any cause, recurrent myocardial infarction or hospitalization for heart failure at 3 year follow-up.
- Secondary end points:
- Individual components of primary endpoint at 6 and 12 months
- Cardiac death
- Stroke
- Transient ischaemic attack
- Major bleeding
- Unstable angina
- Unplanned angiography and revascularization
- Stent thrombosis
Results:
- Mean age 63 ±11 years
- 78% male in both arms
- Smoking rates higher in iFR group
- Majority of patients in both arms had normal or mild LV dysfunction
- Baseline angiographic variables well balanced
- 1 in ten patients in the MRI group did not undergo an MRI and underwent iFR guided bailout procedure at a median of 34 days
- 8% of patients had a non culprit lesion with ischaemia in the MRI group, versus 44.9% of patients in the iFR arm
- 6% of patients in iFR group and 18.7% of patients in the MRI group underwent non culprit lesion coronary artery PCI.
- Primary end point occurred in 9.3% in the iFR group and 9.8% in the MRI group (hazard ratio, 0.95; 95% confidence interval [CI], 0.65 to 1.40, p=0.81)
- Death from any cause or recurrent myocardial infarction did not differ
- Secondary end points similar between groups no difference in adverse events.
- Stent thrombosis was numerically higher in iFR group but was not statistically significant.
Conclusions:
- In patients presenting with STEMI and multivessel coronary artery disease, immediate iFR guided PCI was not superior to a deferred cardiac stress perfusion MRI guided PCI strategy for non culprit lesions.
Limitations:
- Event rate was lower than expected even at 3 years so study may not be powered sufficiently to detect differences in the primary outcome.
- iFR may have been impacted by alterations in microvascular resistance at the time of STEMI, potentially leading to some false positives
- In the MRI group some patients did not undergo additional ischaemia testing or intervention which may also have influenced the trial findings
Comments:
- One fifth of patients in the MRI group did not undergo MRI stress perfusion, of which half underwent bail out iFR guided PCI.
- iFR identified more positive lesions than cardiac stress perfusion MRI and therefore smaller ischaemic territories may have been missed on MRI. Despite this there remained to be no difference in outcomes.
- The number of primary endpoints was lower than predicted so further adequately powered trials need to be performed.
- Although not adequately powered, this trial showed that a deferred strategy is safe in patients presenting with STEMI with multi vessel disease, and equally there is no increase in adverse events or PCI complications in those patients undergoing multi vessel PCI at the time of STEMI.
PROCTOR: PCI of Native Coronary Artery vs Saphenous Vein Graft After Prior Bypass Surgery
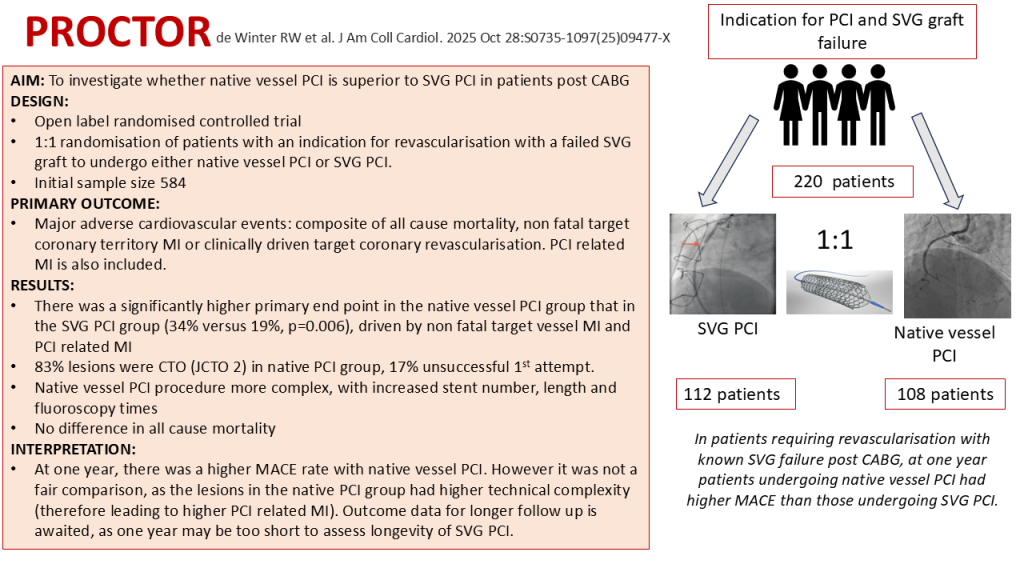
Rationale
- Current guidelines recommend PCI of the native coronary artery instead of the failed vein graft, however this is based on observational data. There are challenges with both approaches: vein graft PCI has a high risk of no reflow and early stent failure and native vessel PCI often involves extensive atheroma and high rates of CTO given the aggressive nature of post bypass native coronary artery disease. The aim of this trial was to compare the outcomes of native vessel PCI versus SVG PCI in post CABG patients.
Publications details:
- de Winter RW et al. PROCTOR Trial Investigators. PCI of Native Coronary Artery vs Saphenous Vein Graft After Prior Bypass Surgery: A Multicenter, Randomized Trial. J Am Coll Cardiol. 2025 Oct 28:S0735-1097(25)09477-X. doi: 10.1016/j.jacc.2025.09.1577. Epub ahead of print. PMID: 41159978
Study design
- International open label, randomised controlled trial
- 1:1 randomisation of patients with a heart team defined indication for percutaneous revascularisation to undergo PCI to the vein graft or PCI to the native vessel.
- Successful PCI defined as residual stenosis less than 30% with restoration of TIMI 3 flow in distal vascular bed of the bypassed native vessel.
- If initial PCI strategy failed, second attempt was performed or cross over to the alternative strategy.
- Intention to treat analysis
- 1, 3 and 5 year follow up planned
Trial Information
- 341 patients screened, 220 patients randomised
- 14 centres in Europe
- Initial sample size 584 patients
- Follow up during index hospitalization and one year clinical outcomes were assessed through national registry databases, longitudinal review of electronic medical records and telephone interviews
- Recruitment between December 2018 and December 2023
Inclusion
- 18 years or older
- Significant diameter stenosis (>50%) in an SVG in invasive coronary angiography
- Revascularisation clinically indicated by local heart team
- Both SVG and native vessel were technically treatable via PCI.
Exclusion
- Cardiogenic shock
- STEMI
- Ongoing ischaemia in setting of NSTEMI
- Estimated life expectancy of less than 3 years
- Unfavourable angiographic characteristics: target vessel diameter <2.5mm, graft diameter >5.5mm, marked aneurysmal degeneration of the bypass graft, heavy thrombus burden
- Early graft failure
Outcomes
- Primary outcome: major adverse cardiovascular events (MACE): composite of all cause mortality, non fatal target coronary territory MI or clinically driven target coronary revascularisation. PCI related MI occurring during index procedure was included.
- Target vessel failure
- Target lesion revascularisation
- Renal failure requiring RRT
- Patient reported quality of life
Results:
- 220 patient randomly assigned to undergo native PCI (n=108) or SVG PCI (n=112)
- 10 patients had died within 1 year of index procedure.
- Mean age 73 ± 7, 84% male
- Mean time between CABG and index PCI was 18± 8 years
- Baseline characteristics well balanced
- Out of the 112 in the SVG PCI group, 3 crossed over to native vessel PCI. Out of 108 patients in the native vessel PCI group, 12 crossed over to SVG PCI.
- 1/5th of patients had prior target SVG PCI
- Numerically higher numbers of ACS in native vessel PCI group
- Target lesion in the native PCI group was a CTO in 83% of patients
- Patients in native PCI group had higher numbers of stents implanted (7 ± 1.3 vs 1.3 ± 0.8; P< 0.001), longer total stent length (p<0.001), fluoroscopy time (p<0.001) and contrast volume (p<0.001)
- In native vessel PCI group, initial procedure not successful in 17%, of which 11% if these crossed over, and those 7 remaining only 2 had successful PCI of native vessel.
- In SVG PCI group 10% were not successful, of which 3% had PCI of native vessel and 7% managed conservatively.
- Final technical success similar in both groups, above 90%.
- J CTO score in both groups were 2, with half of patients had retrograde dissection re-entry as crossing technique
- At 1 year follow up, composite end point occurred in 34% of patients in the native PCI group and 19% of patients in SVG OPCI group (HR: 2.14; 95% CI: 1.25-3.65; P =006)
- No difference in mortality
- Difference driven by non fatal target coronary territory MI and also PCI related MI
- Similar differences seen in the as treated analysis.
- No interaction observed between treatment effect and age, sex, diabetes, presence of native vessel CTO and clinical presentation with ACS
Conclusions:
- In patients post CABG with an indication for revascularisation, a strategy of PCI to the SVG was associated with lower MACE as compared with a native vessel PCI approach.
Limitations:
- Small patient numbers due to early termination
- Slow recruitment (3 patients per centre per year) with a risk of selection bias
- Lack of power most important when interpreting secondary end points
- Decisions regarding repeat revascularisation were down to the treating physician.
- Noa medical therapy arm included.
Comments:
- First randomised controlled trial asking this important question.
- The results of this trial go against the widely held belief that native vessel PCI is preferable to SVG PCI.
- Whilst a heart team was involved in ensuring technical feasibility of both arms in this trial, a high proportion of patients in the native vessel PCI arm had native vessel CTOs classified as difficult with more than 50% requiring a retrograde approach. Therefore, procedural complexity was higher, with longer stents, increased fluoroscopy time and contrast volume in the native PCI group. This may have led to higher PCI related MI.
- Graft occlusion post native vessel PCI was not routinely performed, and this competitive flow may have also played a role in the increase in events seen in the native CI group.
- MACE rates were higher in the native vessel PCI group as compared to other CTO trials, which in part may be reflective of the post CABG population which are often of higher risk due to their co-morbidities and complexity of disease.
- Recruitment was slow (average 3 patients per centre per year), and so the trial was terminated before the total number of patients were recruited. This means that the secondary end points should be interpreted with a caution.
- One year may be too short to assess for outcomes post SVG PCI, and it will be interesting to see if the SVG PCI arm catches up with the native PCI in terms of adverse outcomes with longer term follow up.
- Currently with the current evidence base, decisions regarding whether to PCI an SVG or native vessel should be assessed on a case by case basis, taking into account factors such as lesion complexity and the presence of prior SVG graft failure.
- There were higher numerical rates of stabilised ACS in the native PCI group and low use of embolic protection in SVG group, to suggest these patients overall may have been in a lower risk category.










